Fission Reactions Can Get Out of Control Without What Taking Place?
Fission vs Fusion
Nuclear fusion and nuclear fission are two dissimilar types of energy-releasing reactions in which energy is released from high-powered diminutive bonds between the particles within the nucleus.
The main departure between these 2 processes is that fission is the splitting of an atom into two or more smaller ones while fusion is the fusing of two or more smaller atoms into a larger one.
Nuclear Fission
- Fission reaction does non normally occur in nature
- Fission produces many highly radioactive particles
- The energy released by fission is a 1000000 times greater than that released in chemical reactions; merely lower than the free energy released past nuclear fusion
-
One class of nuclear weapon is a fission bomb, also known equally an atomic bomb or atom bomb
-
Fission is the splitting of a large cantlet into two or more smaller ones
-
Critical mass of the substance and high-speed neutrons are required
-
Takes piddling energy to split two atoms in a fission reaction
-
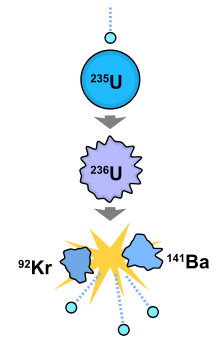
Nuclear fission is the splitting of a massive nucleus into photons in the course of gamma rays, complimentary neutrons, and other subatomic particles. In a typical nuclear reaction involving 235U and a neutron:
23592U + n = 23692U followed past 23692U = 14456Ba + 89 36Kr + 3n + 177 MeV
Nuclear Fusion
-
Fusion occurs in stars, such every bit the sun
-
Few radioactive particles are produced by fusion reaction, just if a fission "trigger" is used, radioactive particles will upshot from that
-
The energy released by fusion is 3 to four times greater than the energy released by fission
-
One class of nuclear weapon is the hydrogen bomb, which uses a fission reaction to "trigger" a fusion reaction
-
Fusion is the fusing of 2 or more lighter atoms into a larger 1
-
High density, high temperature environs is required
-
Extremely high free energy is required to bring two or more protons close plenty that nuclear forces overcome their electrostatic repulsion
-
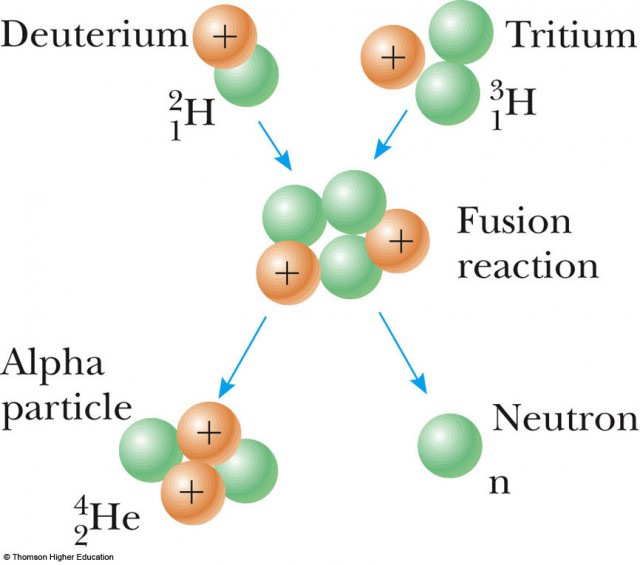
Nuclear fusion is the reaction in which two or more nuclei combine together to form a new chemical element with college atomic number (more than protons in the nucleus). The free energy released in fusion is related to E = mc^2 (Einstein'southward famous energy-mass equation). On earth, the about likely fusion reaction is Deuterium–Tritium reaction. Deuterium and Tritium are both isotopes of hydrogen. 2 1Deuterium + three 1Tritium = 42He + 10n + 17.6 MeV
-
Fusion of deuterium with tritium creating helium-4, freeing a neutron, and releasing 17.59 MeV of free energy
Advantage of Nuclear Fusion over Nuclear Fission
- In case of fusion reactions, fusion reactors cannot sustain a chain reaction so they can never melt down like fission reactors
- Fusion reaction produces very less or, if the right atoms are chosen, no radioactive waste
- In case of nuclear fission big nuclear waste is produced and disposal of radioactive waste is a complicated trouble
- For nuclear power, fusion is the better pick
-
The energy released by fusion is three to 4 times greater than the energy released past fission. This is considering the amount of mass transformed into energy is that much greater in a fusion reaction than in a fission reaction
-
Fusion is essentially inexhaustible, depression-cost fuel, bachelor worldwide
-
High free energy-density of fuel in fusion, allowing straightforward base of operations-load ability product without major transportation cost
-
In fusion at that place is no product of greenhouse gas, soot or acid rain, and no possibility of runaway reaction or meltdown that could pose a risk to public rubber with minimal proliferation risk
-
Fusion has but brusque-lived radioactive wastes
References:
Published at : Updated
greshamwitheniand.blogspot.com
Source: https://research.binus.ac.id/rigpcs/2013/12/06/fission-vs-fusion/
0 Response to "Fission Reactions Can Get Out of Control Without What Taking Place?"
Post a Comment